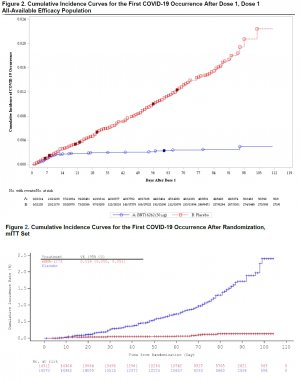
There is no vaccine that is 100% effective and there are a multitude of factors and variables that you cannot draw any specific conclusions. You also need to look at the outcomes and severity of infections in the treated vs. placebo, and there was a significant difference. In the Pfizer trial, the few individuals that did develop COVID did not require hospitalization vs. some on placebo.
Normally, and this may be the case in other countries more so than the US, there are post marketing surveillance reporting which looks at adverse effects (serious) to try to determine side effects (like anaphylaxis) that occur in very low frequency that your sample size needs to be in the millions and if further specific recommendations can be made. When you look at the trial data for both the Pfizer and Moderna vaccines they do what is called an intent to treat analysis, which in general is that if you received treatment, 1 or more doses you are counted in the analysis and often if you are lost to follow-up that is considered an endpoint or failure. It varies on how the data is reported and analyzed, but the primary and secondary endpoints are set before the trial starts. We are also dealing with a virus that has a high mutation rate and selective pressures will cause it to mutate quickly. To say that a vaccine will be effective against X number of mutations, I do not know of any science that supports that theory and probably just statistically modeling that can be anything you want it to be.
Data on COVID incidence for the Pfizer and Moderna studies as aggregate data from Dose 1.
The US ranks very poorly in having a systemic plan and guidance for who gets vaccinated, distribution, priority, notification, etc. The CDC has set guidelines but the states seem to do it all differently in each state and most healthcare providers in the community do not have a clue of how and when. The current guidelines are:
CDC recommends giving COVID-19 vaccine in phases:
1a: Healthcare personnel and Long-term care facility residents
1b: Frontline essential workers and People age 75 years and older
1c: People aged 65 through 74 years and People aged 16 through 64 years with underlying medical conditions and Other essential workers
COVID-19 vaccines protect against COVID-19. Get safety info and more.

www.cdc.gov
The 1a ranks Healthcare personnel and Long-term care facility residents in the same rollout. There may be some logistical issues with the cold storage and distribution that requires distribution points to be major hospitals st this point. The Moderna product is a bit more easily stored and probably accounts for the differences in who is receiving one or the other vaccine. As far as the option to be vaccinated or not, that is an individual right, but employer's may have specific guidelines depending on risk to other's that one may be in contact with. There is also the question of determining previous history of COVID.
As far as the COVID testing kits and positivity, this is a very murky area, in that we talk about specificity and sensitivity of the tests, they vary widely and also when one becomes positive. The current PCR type are the most specific and sensitive, ironically many people that test positive are after the infectious period of the virus. They are still useful as a trend to the of the virus in the population, but clearly only represent a fraction of the total number of individuals that have had COVID, in particular when you factor in the number of asymptomatic individuals. Having had previous exposure to COVID, and survived, does not tell/factor in the risk for a recurrence from a different mutation and also it is unclear as to how this will compare to those vaccinated. If you look at the current CDC recommendations for individuals that had a previous history of Herpes Zoster, they still recommend individuals to get the Shingrix vaccine which is 97% effective at preventing herpes zoster (shingles) in folks over 50. I have run a number of these clinical trials and often you would see shingles recurrence in those not vaccinated.
CDC recommends a single dose of herpes zoster vaccine for people 60 years old or older. Zostavax vaccine is approved by FDA for people age 50 years and older. However, CDC does not recommend routine use of herpes zoster vaccine in people age 50 through 59 years.

www.cdc.gov
So it is all a matter of choice, worthwhile to know that at least there are effective vaccines available and that the vaccination programs have started. It is helpful for those who have been vaccinated to share their experience positive or negative.